“Continuous” and “one-shot” serratus plane block in uniportal video-assisted thoracic surgery: preliminary results
Introduction
Technological development has led thoracic surgery to a deep revolution in terms of minimally invasive techniques. This resulted in a reduction of the dimensions of the surgical access and a consequent reduction of the duration of the post-operative hospitalization and of its psycho-physical impact on the patient.
In fact, in the past, thoracic surgery was included among the most painful surgeries, characterized by long post-operative hospitalizations (7–10 days) and weighty therapies based on poly-pharmacological schemes of pain-killers. In the last years, we have moved on to a less invasive approach, with video-assisted surgery based on three, two or only one trans-thoracic accesses.
Thoracic surgery performed with a minimally invasive “uniportal video-assisted thoracic surgery” (U-VATS) technique is one of the most innovative procedures and permits to perform the entire surgery using a single muscular-sparing incision of 3–4 cm (1). This technique allows an important reduction of post-operative pain in comparison to a thoracotomy, but it doesn’t avoid it (2) completely. Therefore, management of post-operative pain remains a fundamental theme in thoracic surgery: there is a common use of intravenous systemic pain killers and other different antalgic techniques aiming at blocking the painful transmission of the operated hemithorax. These techniques, though, sometimes bring on many side effects and, for this reason, need to be carried out by well-trained specialists (3,4).
This issue has led to a research for alternative techniques: in 2013, Blanco et al. described the “serratus plane block” (SPB) (5). In the following years, other articles described the efficacy of the same technique associated with mini-invasive thoracic surgery with multiple accesses (6,7).
We report our preliminary experience about the application of the “SPB”, for the first time in U-VATS surgery.
Methods
We present clinical data of 44 patients who underwent elective U-VATS surgery and subsequent post-surgical pain treatment through “SPB” between March 2018 and December 2018 at the Department of Thoracic Surgery of Fondazione Policlinico Universitario “A. Gemelli” IRCCS (Rome, Italy).
To minimize confounding factors in the evaluation of analgesic efficacy, we involved patients not presenting the characteristics summarized in Table 1.
Table 1
| Absence of informed consent to surgery and treatment of clinical data |
| Previous chemotherapy treatment |
| Age <18 years |
| Epilepsy |
| Diabetes mellitus |
| Preoperative VAS for subjective pain assessment >0 |
| Preoperative episodic VAS (cough pain) >0 |
| Allergy to local anesthetics and pain medication (NSAIDs and opioid derivatives) |
| Chronic pain-relieving therapy |
| Chronic intake of gabapentinoids (gabapentin, pregabalin) or other antiepileptic/anticonvulsant drugs |
| Abuse of alcohol and drugs |
| Previous thoracic surgery |
| Intensive postoperative monitoring |
NSAIDs, nonsteroidal anti-inflammatory drugs; VAS, visual analog scale.
The preoperative routine (blood tests, ECG, respiratory function test) was performed on all patients. Prior to surgery, visual analog pain scale (VAS) was submitted to all patients: “0” corresponded to no pain and “10” to the worst possible pain.
The surgical procedure
Thoracic surgery was performed through a minimally invasive “U-VATS” technique using a single muscular-sparing incision of 3–4 cm at the 5th intercostal space on the mid-anterior axillary line, without costal divarication. In this technique, a wound protector facilitates the introduction of thoracoscopic instruments and preserves the intercostal nerve from the continuous stress of surgery.
Possible surgical interventions on our patients included anatomical (segmentectomy, lobectomy) or non-anatomical (wedge) pulmonary resection, both for benign and malignant pathology.
SPB technique in U-VATS
Soon after surgery, SPB analgesic technique was applied.
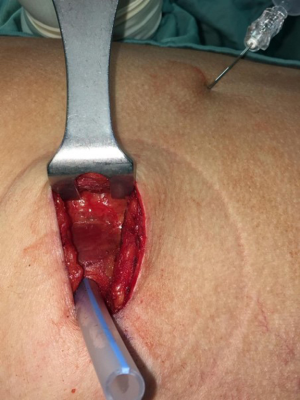
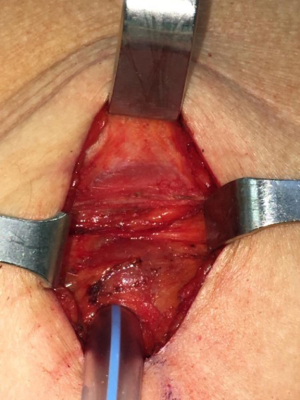
The anterior edge of the dorsal muscle represents the posterior limit of the uniportal access: during the preparation of the surgical access, a “pocket” is created between the lower fascia of the dorsal muscle and the upper fascia of the serratus muscle (Figure 1), allowing an easy “muscle sparing” incision of the serratus and the realization of a softer superior margin of the surgical access.
Routinely, all patients at the end of the surgical time are subjected to an intercostal anesthetic block with 20 mL of Ropivacaine at a concentration of 5 mg/mL (injections at the level of: surgical access space, 2 intercostal spaces superiorly and 2 intercostal spaces inferiorly, with 4 mL of anesthetic for each intercostal space).
The successive SPB consisted in the administration of local anesthetic between the superficial fascia of the serratus muscle and the lower fascia of the dorsal muscle. This anatomical area permits the creation of a plane on which the anesthetic “film”, depending on the quantity of the administered anesthetic, can distribute itself on a cranio-caudal direction potentially going from T2 to T9 metamers and, on an antero-posterior direction, from the anterior axillary line to the posterior axillary line. This distribution allows a prolonged analgesia of the operated hemithorax through the blockage of the superficial branches of the intercostal nerves belonging to the T2-T9 metamers and of the nerves coming from the cervical plexus (C5-C6), including the dorsal nerve and the long thoracic nerve, that result among the co-responsible factors for post-operative pain in thoracic surgery.
SPB was then conducted in two different ways: in a first group of 22 consecutive patients a “continuous” SPB (cSPB) was performed, while a “one-shot” SPB (osSPB) was performed in the second group of other 22 patients.
cSPB and osSPB technique in U-VATS
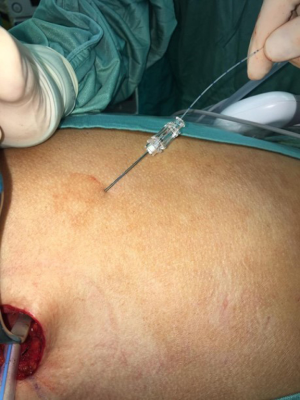
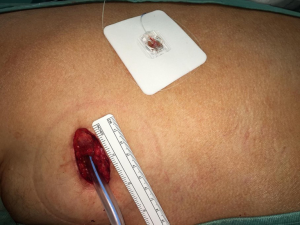
A cSPB consists in placing, under visual guidance, an epidural-like catheter (Smiths medical Portex Epidural Minipack System 1 with clamp, 18 Gauge) between the lower fascia of the dorsal muscle and the upper fascia of the serratus muscle with a cutaneous exit near to the surgical access (Figures 2,3). At the end of the synthesis of the surgical access, before the extubation of the patient, a “priming” infiltration of 20 mL of Ropivacaine is carried out from the catheter at a concentration of 2 mg/mL (total of 40 mg of Ropivacaine) and the catheter is connected with an elastomer (Figure 4) with an infusion rate of 5 mL/h (total volume 270 mL) for the local administration of Ropivacaine at a concentration of 2 mg/mL (10 mg/h).
The osSPB procedure, instead, is carried out, after closing the fascia of the serratus muscle, by sliding a flushing needle posteriorly the uniportal access on the anterior fascia of the serratus and under the dorsal muscle for about 5 cm and performing an infiltration of 20 mL of Ropivacaine at a concentration of 2 mg/mL (total of 40 mg of Ropivacaine).
The total dose of Ropivacaine in both procedures never exceeds the dosage of 3 mg/kg.
Routine post-operative analgesic therapy administration
Painkiller therapeutic schedule set in the post-operative days was the following in all patients:
- Paracetamol 1,000 mg every 8 hours;
- NSAIDs (Ketorolac 30 mg) in case of VAS >4 (maximum dosage every 8 hours);
- Opioid derivates (Tramadol 50 mg/Oxycontin 10 mg) in case of VAS >4 as an alternative to Ketorolac (maximum dosage every 6 hours).
Pain assessment
Measurements based on VAS were performed as follows:
- 8 hours after the end of the surgical procedure;
- at 8 am on the first, second and third post-operative day (POD);
- at 8 pm on the first, second and third POD;
- episodic cough pain on the first, second and third POD;
- 2 hours after drainage removal.
Statistical analysis
Statistical analysis was performed using PASW Statistics for Windows, Version 18.0 (SPSS Inc., Chicago, IL, USA). Continuous variables were expressed as mean and standard deviation. Pearson χ2 test and Fischer’s exact test were used to compare discrete variables and Student’s t-test to compare means between two continuous variables. A P value less than 0.05 was considered statistically significant.
Results
Clinical data of 44 patients, whose characteristics are summarized in Table 2, were analyzed.
Table 2
| Patients | cSPB | osSPB |
|---|---|---|
| Number | 22 (50%) | 22 (50%) |
| Sex (male) | 10 (45.5%) | 10 (45.5%) |
| Age (years) | 63.3 (±12.0) | 67.8 (±10.0) |
| Side (right) | 7 (31.8%) | 8 (36.4%) |
| Length of postoperative stay (days) | 3.9 (±0.9) | 3.8 (±1.0) |
cSPB, continuous serratus plane block; osSPB, one shot serratus plane block.
Twenty-two (50%) of them underwent cSPB and 22 (50%) of them the osSPB procedure. Both groups had 10 (45%) male and 12 female (55%) patients.
Types and distribution of the surgical procedures undertaken by the patients in the two groups are described in Table 3.
Table 3
| Group | Lobectomy | Segmentectomy | Wedge resection |
|---|---|---|---|
| cSPB (number) | 12 (54.5%) | 2 (9.1%) | 8 (36.4%) |
| osSPB (number) | 11 (50.0%) | 2 (9.1%) | 9 (40.9%) |
| Total | 23 (52.3%) | 4 (9.1%) | 17 (38.6%) |
cSPB, continuous serratus plane block; osSPB, one shot serratus plane block.
Among the population analyzed, there were no cases of major postoperative complications or minor complications like pneumonia or atelectasis, with the need of bronchoaspiration.
In the osSPB group, administration of the anesthetic took a few seconds, while in the cSPB group, post-surgical mean time placement of the catheter was 282±39 s.
Post-operative pain assessment derived from the administration of the VAS scale in the two groups is reported in Table 4.
Table 4
| Group | Po pain | I day morning | I day episodic | I day evening | II day morning | II day episodic | II day evening | III day morning | III day episodic | After drainage removal |
|---|---|---|---|---|---|---|---|---|---|---|
| cSPB (VAS ± SD) | 2.63±1.62 | 2.72±1.63 | 3.86±1.76 | 2.81±1.67 | 3.09±1.59 | 4.04±1.64 | 2.72±1.57 | 2.54±1.64 | 3.59±1.58 | 3.18±1.11 |
| osSPB (VAS ± SD) | 2.63±0.93 | 4.22±1.28 | 5.54±1.37 | 4.72±1.42 | 4.5±1.5 | 5.72±1.54 | 4.27±1.6 | 4.09±1.47 | 5.54±1.67 | 4.04±1.52 |
| P value | 1 | 0.002 | 0.001 | 0.001 | 0.005 | 0.001 | 0.003 | 0.003 | 0.001 | 0.042 |
cSPB, continuous serratus plane block; osSPB, one shot serratus plane block; Po, post-operative; VAS ± SD, visual analog scale ± standard deviation.
No significant difference between the two groups was found in postoperative pain intensity assessed at 8 hours after surgery, while there was a statistical significance since the first POD for patients in the cSPB group, who had better pain control compared with the osSBP group.
Analyzing the differences in the type of additional post-operative painkillers required in case of VAS >4, we found that patients who underwent cSPB required, in comparison to osSPB group, less frequently both NSAIDs (1.08/die vs. 1.86/die) and opioid derivates (0.056/die vs. 0.42/die) (Table 5). We also observed that even in the routine post-operative analgesic therapy administration, some of the patients in both groups refused paracetamol as they didn’t have pain.
Table 5
| Pain killers (daily required administrations) | Paracetamol | NSAIDs | Tramadol | Oxycontin |
|---|---|---|---|---|
| cSPB group | 2.63 (±0.48)/die | 1.08 (±0.78)/die | 0.056 (±0.21)/die | 0.045 (±0.2)/die |
| osSPB group | 2.86 (±0.34)/die | 1.86 (±0.34)/die | 0.42 (±0.52)/die | 0.75 (±0.77)/die |
| P value | 0.085 | 0.001 | 0.005 | 0.001 |
cSPB, continuous serratus plane block; osSPB, one shot serratus plane block; NSAIDs, nonsteroidal anti-inflammatory drugs.
Discussion
Post-operative pain management remains a fundamental topic in thoracic surgery.
Punctual and optimal post-operative pain control is an important determinant for the quality of recovery and life of the patient, as it allows an early mobilization of the patient and an effective cough to expel secretions, preventing the development of respiratory complications such as pneumonia and atelectasis (8,9).
In the post-surgical setting, there is a common use of intravenous systemic pain relievers such as anti-inflammatory drugs (NSAIDs) and opioid derivates, which may however have adverse effects such as respiratory depression, itching, sedation, nausea, vomiting, and, in some cases, addiction phenomena (10). Other commonly used antalgic techniques aiming at blocking the painful transmission of the operated hemithorax include the epidural block, the paravertebral block and the intra/extrapleural block (11,12) which allow a blockage of the deep intercostal nerves and of their superficial branches. However, these maneuvers must be performed by a well-trained specialist, as important side effects such as hypotension (4.6%), vascular puncture (3.8%), pleural puncture (1.1%) and pneumothorax (0.5%) may occur (13,14).
Pain in uniportal thoracic surgery is caused by the surgical wounding of the serratus muscle and by the positioning and presence of a post-surgical drainage. Such pain is lower than in thoracotomy although the continuous movement of the drainage through the surgical wound can generate pain.
The application of a central analgesic blocks, such as the paravertebral and epidural ones, to U-VATS would become a disproportionate choice when considering their possible side effects. Therefore, the idea of applying the SPB to uniportal surgeries derives from the necessity to precociously and preventively anesthetize the operated hemithorax so to avoid the onset of pain caused by the rapid mobilization and autonomization of the patient, without fearing excessive side effects.
The SPB technique described by Blanco in 2013 allows having a prolonged analgesia of the operated hemithorax because the relatively low vascularization of this anatomic plane allows a film-like distribution and slow absorption of the local anesthetic. Its analgesic effect is mediated by the blockage of the lateral cutaneous branches of the intercostal nerves and consequently by the retrograde block of the deep intercostal branches. A further analgesic effect seems to be carried out by the direct block of the long thoracic nerve, which runs superficially to the serratus muscle, and of the thoraco-dorsal nerve, both originating from the brachial plexus (15).
As described, the SPB allows the direct block of three pain components (superficial branches of intercostal nerve, long thoracic nerve, thoraco-dorsal nerve) coming from two different districts (the brachial plexus and thoracic spinal nerves) and the retrograde block of a fourth pain component (the deep intercostal nerve).
Nowadays, the effectiveness of the SPB in both open and thoracoscopic surgery is being demonstrated in several studies and no major side effects have been described up to now.
Ökmen et al. (16) demonstrated that the use of SPB decreases the use of opioids in the immediate post-operative period in patients undergoing thoracotomy.
Saad et al. (11) compared the use of SPB and paravertebral block before surgical incision in patients undergoing thoracotomy, demonstrating that both provided adequate analgesia for the first 24 hours with an equivalent level of analgesia of the two techniques for the first 9 hours. Thoracic paravertebral block has documented better pain control compared to the SPB from the 9th to the 24th hour.
Park et al. (17) demonstrated that a pre-operative serratus anterior plane block with ropivacaine reduces fentanyl consumption in the first 24 hours after thoracoscopic surgery compared with no block.
Semyonov et al. (18) concluded that SPB, compared with intravenous opioid administration and NSAIDs, was an effective adjuvant treatment option for post-thoracoscopic surgery pain relief and it has some significant merits, particularly its ease of use and its low potential of side effects.
Kim et al. (19), in a triple-blind randomized study, demonstrated how the use of SPB in patients undergoing VATS surgery improves the analgesia and the quality of recovery for 2 days during the early postoperative period. In literature there are no studies concerning the use of the SPB in thoracic surgery performed with the U-VATS technique. Between March 2018 and December 2018, we applied the SPB technique to 44 patients undergoing elective uniportal thoracic surgery. We applied it in a one-shot modality in 22 patients and through a catheter in a continuous modality in other 22 patients.
It is already well known that minimally invasive surgical interventions, especially from a single access, are correlated with a reduced post-operative pain and a reduced consumption of pain-killers, both for pulmonary surgery (20) and for esophageal surgery (21). With the application of a SPB, we are able to guarantee our patients an even less painful experience, with less complications, early mobilization and consequent discharge.
In our study, analysis of post-operative pain intensity at different timings after surgery showed that cSPB was comparable to osSPB in the first 8 hours after surgery, but it became significantly advantageous in the following hours of the first, second and third POD and after drainage removal (Table 4).
The continuous application of this technique has several advantages:
- there is a more specific and local pain management: the source of pain is locally treated and there is a lower final quantity of administered analgesic drugs; this characteristic meets the gold standard of pain management for anesthesiologists (22-24);
- there is a lower request for systemic analgesic therapy administration: patients are comfortable with the cSPB catheter alone and less frequently ask for additional pain treatment;
- with a locally managed pain and a lower administration of NSAIDs and opioid derivates, side effects are brought down to a very little, if significant at all, number of cases (none, in our experience);
- furthermore, the time used to position the cSPB catheter in the operating room does not lengthen the duration of surgery (+4 min) and gets faster with the acquisition of experience by the operator, who doesn’t necessarily have to be an anesthesiologist or an expert, as the procedure is quite easy to be performed.
As a preliminary study, it has some limits, like the small size of the population involved and the absence of a randomization in involving patients. Therefore, more powerful randomized studies are claimed to confirm our early conclusions.
According to our experience, both osSPB and cSPB seem to provide a good level of analgesia for the first 8 hours postoperatively. After 12 hours, however, cSPB provides better pain control and reduced request of pain therapy administration. Both the procedures seem to be safe, with no observed side effects in our series. Accordingly, the combination of U-VATS with cSPB, may offer patients a good level of pain tolerance enhancing the quality of their recovery in the postoperative period.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Shanghai Chest for the series “The Second Roman Tips and Tricks in Thoracic Surgery - the Great Potential of Uniportal VATS”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/shc.2019.07.04). The series “The Second Roman Tips and Tricks in Thoracic Surgery - the Great Potential of Uniportal VATS” was commissioned by the editorial office without any funding or sponsorship. EM served as the unpaid Guest Editor of the series and serves as an unpaid editorial board member of Shanghai Chest from Mar 2018 to Feb 2020. SM served as the unpaid Guest Editor of the series. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Institutional Review Board of Catholic University of Sacred Hearth (Rome) and informed consent was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Ismail M, Swierzy M, Nachira D, et al. Uniportal video-assisted thoracic surgery for major lung resections: pitfalls, tips and tricks. J Thorac Dis 2017;9:885-97. [Crossref] [PubMed]
- Sihoe AD. Uniportal Lung Cancer Surgery: State of the Evidence. Ann Thorac Surg 2019;107:962-72. [Crossref] [PubMed]
- Joshi GP, Bonnet F, Shah R, et al. A systematic review of randomized trials evaluating regional techniques for postthoracotomy analgesia. Anesth Analg 2008;107:1026-40. [Crossref] [PubMed]
- Kavanagh BP, Katz J, Sandler AN. Pain control after thoracic surgery. A review of current techniques. Anesthesiology 1994;81:737-59. [Crossref] [PubMed]
- Blanco R, Parras T, McDonnell JG, et al. Serratus plane block: a novel ultrasound-guided thoracic wall nerve block. Anaesthesia 2013;68:1107-13. [Crossref] [PubMed]
- Madabushi R, Tewari S, Gautam SK, et al. Serratus anterior plane block: a new analgesic technique for post-thoracotomy pain. Pain Physician 2015;18:E421-4. [PubMed]
- Broseta AM, Errando C, De Andrés J, et al. Serratus plane block: the regional analgesia technique for thoracoscopy? Anaesthesia 2015;70:1329-30. [Crossref] [PubMed]
- Elmore B, Nguyen V, Blank R, et al. Pain Management Following Thoracic Surgery. Thorac Surg Clin 2015;25:393-409. [Crossref] [PubMed]
- Piccioni F, Segat M, Falini S, et al. Enhanced recovery pathways in thoracic surgery from Italian VATS Group: perioperative analgesia protocols. J Thorac Dis 2018;10:S555-63. [Crossref] [PubMed]
- Michelet P, Guervilly C, Hélaine A, et al. Adding ketamine to morphine for patient-controlled analgesia after thoracic surgery: influence on morphine consumption, respiratory function, and nocturnal desaturation. Br J Anaesth 2007;99:396-403. [Crossref] [PubMed]
- Saad FS, El Baradie SY, Abdel Aliem MA, et al. Ultrasound-guided serratus anterior plane block versus thoracic paravertebral block for perioperative analgesia in thoracotomy. Saudi J Anaesth 2018;12:565-70. [PubMed]
- Kotzé A, Scally A, Howell S. Efficacy and safety of different techniques of paravertebral block for analgesia after thoracotomy: a systematic review and metaregression. Br J Anaesth 2009;103:626-36. [Crossref] [PubMed]
- Tighe SQ, Greene MD, Rajadurai N. Paravertebral block. Contin Educ Anaesth Crit Care Pain 2010;10:133-7. [Crossref]
- Coveney E, Weltz CR, Greengrass R, et al. Use of paravertebral block anesthesia in the surgical management of breast cancer: Experience in 156 cases. Ann Surg 1998;227:496-501. [Crossref] [PubMed]
- Mayes J, Davison E, Panahi P, et al. An anatomical evaluation of the serratus anterior plane block. Anaesthesia 2016;71:1064-9. [Crossref] [PubMed]
- Ökmen K, Ökmen BM. The efficacy of serratus anterior plane block in analgesia for thoracotomy: a retrospective study. J Anesth 2017;31:579-85. [Crossref] [PubMed]
- Park MH, Kim JA, Ahn HJ, et al. A randomised trial of serratus anterior plane block for analgesia after thoracoscopic surgery. Anaesthesia 2018;73:1260-4. [Crossref] [PubMed]
- Semyonov M, Fedorina E, Grinshpun J, et al. Ultrasound-guided serratus anterior plane block for analgesia after thoracic surgery. J Pain Res 2019;12:953-60. [Crossref] [PubMed]
- Kim DH, Oh YJ, Lee JG, et al. Efficacy of Ultrasound-Guided Serratus Plane Block on Postoperative Quality of Recovery and Analgesia After Video-Assisted Thoracic Surgery: A Randomized, Triple-Blind, Placebo-Controlled Study. Anesth Analg 2018;126:1353-61. [Crossref] [PubMed]
- Louis SG, Gibson WJ, King CL, et al. Uniportal video-assisted thoracoscopic surgery (VATS) technique is associated with decreased narcotic usage over traditional VATS lobectomy. J Vis Surg 2017;3:117. [Crossref] [PubMed]
- Nachira D, Meacci E, Mastromarino MG, et al. Initial experience with uniportal video-assisted thoracic surgery esophagectomy. J Thorac Dis 2018;10:S3686-95. [Crossref] [PubMed]
- Savoia G, Ambrosio F, Paoletti F, et al. SIAARTI recommendations for the treatment of postoperative pain. Minerva Anestesiol 2002;68:735-50. [PubMed]
- Savoia G, Alampi D, Amantea B, et al. Postoperative pain treatment SIAARTI Recommendations 2010. Short version. Minerva Anestesiol 2010;76:657-67. [PubMed]
- Chou R, Gordon DB, de Leon-Casasola OA, et al. Management of Postoperative Pain: A Clinical Practice Guideline From the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists' Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain 2016;17:131-57. [Crossref] [PubMed]
Cite this article as: Ferretti GM, Zanfrini E, Pogliani L, Nachira D, Triumbari EKA, Iaffaldano AG, Gualtieri E, Congedo MT, Meacci E, Margaritora S. “Continuous” and “one-shot” serratus plane block in uniportal video-assisted thoracic surgery: preliminary results. Shanghai Chest 2019;3:39.